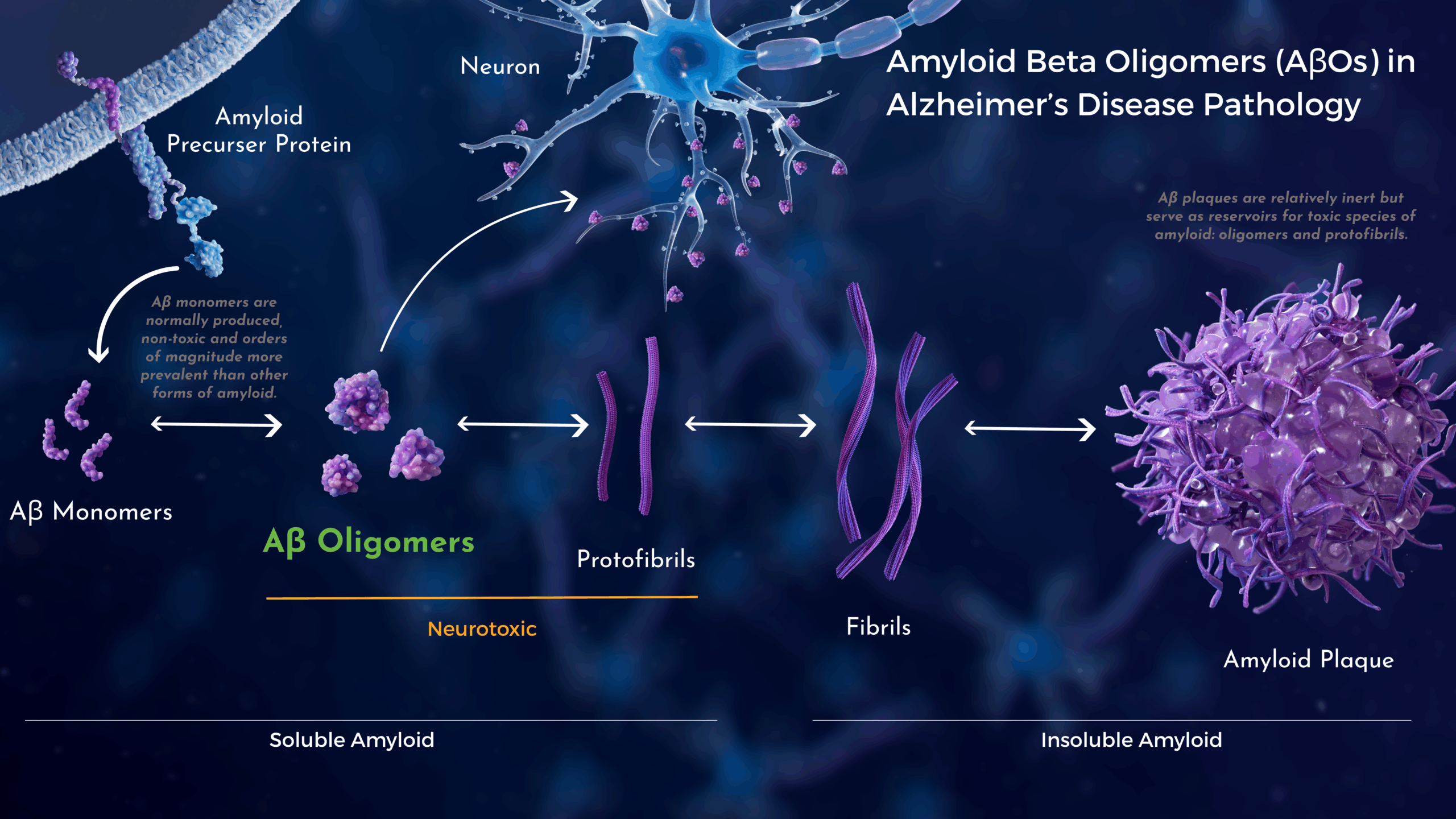
Amyloid Beta (Aβ) Species in Alzheimer’s Disease
In patients with Alzheimer’s disease, five dominant pools of Aβ species have been identified: monomers, oligomers (AβOs), protofibrils, fibrils, and amyloid plaques.1
Many current and prior drug development efforts have been primarily focused on Aβ monomers and amyloid plaques.2,3
Focusing on Toxic AβOs
Acumen’s therapeutic target for Alzheimer’s disease is different. Decades of research support the hypothesis that soluble AβOs are a highly toxic form of Aβ. AβOs are an early trigger and persistent driver of Alzheimer’s-associated pathology and neurodegeneration.
Experimentally, AβOs have been associated with synapse deterioration, tau hyper-phosphorylation, and inflammation. In addition, they have been shown to disrupt brain circuitry.4,5
Recent studies suggest that eliminating the toxic soluble species from the neuronal system may stabilize damaged circuits and slow the rate of decline.3
TARGETING TOXIC AβOS WITH SABIRNETUG (ACU193)
Enter sabirnetug, an anti-AβO monoclonal antibody (mAb) developed to selectively target and inhibit toxic soluble AβOs, preventing them from binding to synapses, and thereby preserving neuronal function. This mechanism of action gives sabirnetug the potential to offer best-in-class efficacy and safety as a next-generation treatment for early Alzheimer’s disease.1,3
In the Phase 1 INTERCEPT-AD study, a novel target engagement assay measured sabirnetug-AβO complexes in cerebrospinal fluid. This assay demonstrated that sabirnetug could cross the blood-brain barrier and bind to AβOs, suggesting the potential clearance of AβOs from the brain.6 Furthermore, this binding led to a reduction in Tau phosphorylation and neurogranin levels, indicating a normalization of Alzheimer’s disease-related changes. Long-term biomarker changes and their correlation with clinical outcomes will be evaluated in the Phase 2 ALTITUDE-AD study.7
Sabirnetug (ACU193):
The First Immunotherapy Drug Candidate Targeting Toxic Soluble AβOs
Sabirnetug is a monoclonal antibody that is distinguished from other anti-Aβ mAbs for several reasons:
- Highly selective for soluble oligomers, a highly toxic Aβ species; other anti-Aβ mAbs are less selective and/or target different amyloid species, such as monomers, fibrils, or plaques2,3
- Designed to provide improved clinical efficacy with lower rate of amyloid-related imaging abnormalities (ARIA) as compared to anti-plaque mAbs1,2
- Prevents toxic soluble AβOs from binding to synapses, thereby preserving neuronal function2
- Potential for cognitive improvement in addition to slowing disease progression2
REFERENCES
- Siemers E, Hitchcock J, Sundell K, Dean R, Jerecic J, Cline E, Iverson K, Moore J, Edgar C, Manber R, Fuin N, Poppe T, Barton R. ACU193, a Monoclonal Antibody That Selectively Binds Soluble Aß Oligomers: Development Rationale, Phase 1 Trial Design, and Clinical Development Plan. J Prev Alzheimers Dis. 2023;10(1):19-24.
- Krafft GA, Jerecic J, Siemers E, Cline EN. ACU193: An Immunotherapeutic Poised to Test the Amyloid β Oligomer Hypothesis of Alzheimer’s Disease. Front Neurosci. 2022;16:848215.
- Goure WF, Krafft GA, Jerecic J, Hefti F. Targeting the Proper Amyloid-Beta Neuronal Toxins: A Path Forward for Alzheimer’s Disease Immunotherapeutics. Alzheimers Res Ther. 2014;6(4):42.
- Cline EN, Bicca MA, Viola KL, Klein WL. The Amyloid-β Oligomer Hypothesis: Beginning of the Third Decade. J Alzheimers Dis. 2018;64(s1):S567-S610.
- Wang X, Kastanenka KV, Arbel-Ornath M, Commins C, Kuzuya A, Lariviere AJ, Krafft GA, Hefti F, Jerecic J, Bacskai BJ. An Acute Functional Screen Identifies an Effective Antibody Targeting Amyloid-β Oligomers Based on Calcium Imaging. Sci Rep. 2018;8(1):4634.
- Cline E, Moore J, Zhang H, Sethuraman G, Siemers E, Dean R, Jerecic J. ACU193-sAβO Complex Measurement in CSF: Additional Analyses Using a Sensitive Assay of Target Engagement for the sAβO-Selective Antibody ACU193 in INTERCEPT-AD [Poster presentation]. Presented at: 16th Clinical Trials on Alzheimer’s Disease (CTAD); October 24-27, 2023; Boston, MA, United States.
- Cline E. Sabirnetug (ACU193) Lowers CSF Neurogranin & pTau181 Levels in INTERCEPT-AD Study in Early AD [Presentation]. Presented at: AD/PD™ 2024 Alzheimer’s & Parkinson’s Diseases Conference; March 5-9, 2024; Lisbon, Portugal.
SELECTED PUBLICATIONS
- Gong Y, Chang L, Viola KL, Lacor PN, Lambert MP, Finch CE, Krafft GA, Klein WL. Alzheimer’s Disease-Affected Brain: Presence of Oligomeric A Beta Ligands (ADDLs) Suggests a Molecular Basis for Reversible Memory Loss. Proc Natl Acad Sci USA. 2003;100(18):10417-10422.
- Lacor PN, Buniel MC, Chang L, Fernandez SJ, Gong Y, Viola KL, Lambert MP, Velasco PT, Bigio EH, Finch CE, Krafft GA, Klein WL. Synaptic Targeting by Alzheimer’s-Related Amyloid Beta Oligomers. J Neurosci. 2004;24(45):10191-10200.
- Hepler RW, Grimm KM, Nahas DD, Breese R, Dodson EC, Acton P, Keller PM, Yeager M, Wang H, Shughrue P, Kinney G, Joyce JG. Solution State Characterization of Amyloid Beta-Derived Diffusible Ligands. Biochemistry. 2006;45(51):15157-15167.
- De Felice FG, Wu D, Lambert MP, Fernandez SJ, Velasco PT, Lacor PN, Bigio EH, Jerecic J, Acton PJ, Shughrue PJ, Chen-Dodson E, Kinney GG, Klein WL. Alzheimer’s Disease-Type Neuronal Tau Hyperphosphorylation Induced by A Beta Oligomers. Neurobiol Aging. 2008;29(9):1334-47.
- Gaspar RC, Villarreal SA, Bowles N, Hepler RW, Joyce JG, Shughrue PJ. Oligomers of Beta-Amyloid Are Sequestered into and Seed New Plaques in the Brains of an AD Mouse Model. Exp Neurol. 2010;223(2):394-400.
- Shughrue PJ, Acton PJ, Breese RS, Zhao WQ, Chen-Dodson E, Hepler RW, Wolfe AL, Matthews M, Heidecker GJ, Joyce JG, Villarreal SA, Kinney GG. Anti-ADDL Antibodies Differentially Block Oligomer Binding to Hippocampal Neurons. Neurobiol Aging. 2010;31(2):189-202.
- Rammes G, Hasenjäger A, Sroka-Saidi K, Deussing JM, Parsons CG. Therapeutic Significance of NR2B-Containing NMDA Receptors and mGluR5 Metabotropic Glutamate Receptors in Mediating the Synaptotoxic Effects of β-Amyloid Oligomers on Long-Term Potentiation (LTP) in Murine Hippocampal Slices. Neuropharmacology. 2011;60(6):982-990.
- Savage MJ, Kalinina J, Wolfe A, Tugusheva K, Korn R, Cash-Mason T, Maxwell JW, Hatcher NG, Haugabook SJ, Wu G, Howell BJ, Renger JJ, Shughrue PJ, McCampbell A. A Sensitive Aβ Oligomer Assay Discriminates Alzheimer’s and Aged Control Cerebrospinal Fluid. J Neurosci. 2014;34(8):2884-2897.
- Viola KL, Bicca MA, Bebenek AM, Kranz DL, Nandwana V, Waters EA, Haney CR, Lee M, Gupta A, Brahmbhatt Z, Huang W, Chang TT, Peck A, Valdez C, Dravid VP, Klein WL. The Therapeutic and Diagnostic Potential of Amyloid β Oligomers Selective Antibodies to Treat Alzheimer’s Disease. Front Neurosci. 2022;15:768646.